MiSight® Spectacle Lenses are designed to make high efficacy myopia management more accessible to myopic children.†‡2,3 Together, we can take on myopia and change lives.4
Right now, myopia is winning
1 in 2 children born today could become myopic by age 20.§5 But with new treatment options available, you can do more than just correct refractive error.

You can:
Ensure every child is given the best option for their long-term eye health and reduce the risk of permanent sight loss from high myopia.6-9
Maintain the highest ‘duty of care’ standards.10
Improve patient loyalty and retention.11
Introducing MiSight® Spectacle Lenses.
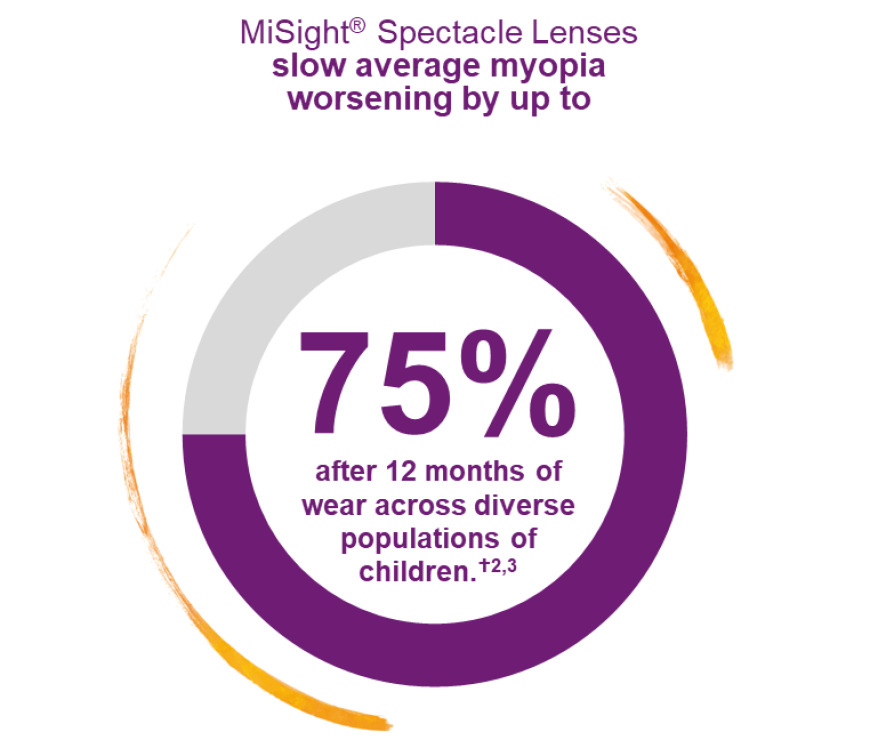
High efficacy for children with myopia.†2,3
MiSight® Spectacle Lenses are designed to help Eye Care Professionals deliver effective, life-changing, myopia management.♢¶1,3,4
Diffusion Optics TechnologyTM
Proven clinical performance for your patients.12-14
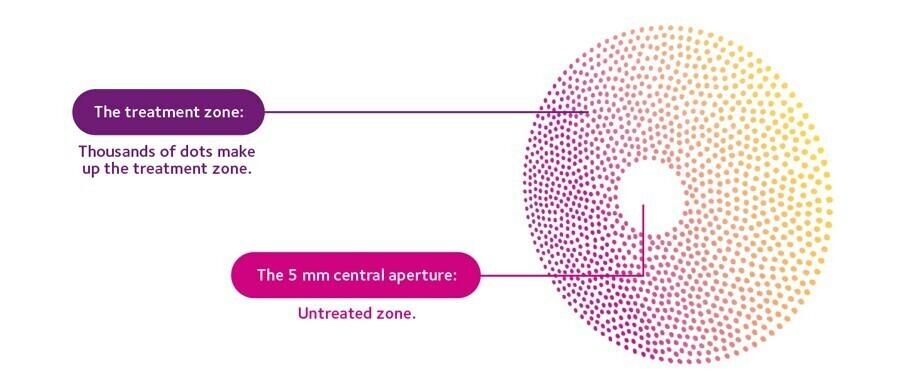
MiSight® Spectacle Lenses are designed to provide contrast-lowering myopia management whilst preserving excellent visual acuity.3

Tried, tested and proven.
Lenses with Diffusion Optics TechnologyTM have been proven to slow myopia in gold-standard clinical trials for myopic children from aged 6 and are available in both tough and thin materials. **15-17
Satisfaction guaranteed.
1. Adaptation guarantee
2. Cosmesis guarantee
3. Prescription change guarantee
MiSight® Spectacle Lenses
Make upgrading easy for you, parents and patients. ††‡


Let’s take on myopia and change lives with MiSight® Spectacle Lenses.4
Contact your CooperVision Territory Manager or Myopia Management Specialist for more details.
| Product Range | Trivex® 1.53 |
| Refractive index | 1.53 |
| Lens blank diameter (mm) | 70 |
| Sphere power (DS) | -0.25 to -8.00 (0.25 steps)* |
| Cylinder power (DC) | -0.25 to -2.00 (0.25 steps)* |
| Abbe value | 45 |
| Optical design | Diffusion Optics Technology™ (DOT) |
* Not available in plano/plano.
* Reduction in progression of spherical equivalent refraction and axial length from baseline over 12 months was 74% and 50%, respectively (p<0.0001).
† Based on a North American patient population aged 6–10 years (least-squared means) and a Chinese patient population aged 6–13 years (unadjusted means). [Significant progression defined as >0.25 D change from baseline.]
‡ Assumes acceptance of MiSight® Spectacle Lenses recommended retail price. Retail pricing is at customer’s discretion and CooperVision do not seek to influence this.
§ Based on the expectation that more than 50% of the world’s population will be affected by myopia by 2050.
♢ No clinically meaningful change in refractive error means that there was less than a 0.25D increase in myopia from baseline as measured by cycloplegic autorefraction (p<0.0001).
¶ Reduction in progression of spherical equivalent refraction and axial length from baseline over 12 months was 74% and 50%, respectively (p<0.0001).
** BJOG defines randomised controlled trials (RCTs) as the “Gold-standard" in clinical trials studying causal relationships between interventions and outcomes.
‡ Assumes acceptance of MiSight® Spectacle Lenses recommended retail price. Retail pricing is at customer’s discretion and CooperVision do not seek to influence this.
†† Dispensing as single vision lens dispensing. Pupil centre height measurements required.
* Not available in plano/plano.
INDICATION: MiSight® Spectacle Lenses are indicated for the correction of refractive ametropia (myopia and/or astigmatism) and for the reduction in the rate of myopic progression in phakic children who are aged 6 to 13 years old with non-diseased eyes. MiSight® Spectacle Lenses should be worn constantly for all activities except for those outlined in WARNINGS, within the legal manufacturer’s Instructions for Use (a minimum of 10 hours per day).
Regulatory indications of and access to interventions for slowing myopia progression vary by country. CooperVision does not endorse off-label prescribing.
References
1. Rappon J, Neitz J, Neitz M, Young G, Chalberg T. CYPRESS 12-month Results: Safety and Efficacy from a Pivotal Study of Novel Spectacle Lenses Designed to Reduce Myopia Progression. Optometry and Vision Science. 2020;97:E-abstract 200036.
2. Laughton D., et al. Control of myopia using diffusion optics spectacle lenses in a Chinese population: 12-month results. 2025 ARVO poster presentation. 24-month, randomized and controlled clinical trial across 5 Chinese hospital sites, evaluating DOT spectacle lenses (n=128) versus control standard single vision spectacle lenses (n=58) in myopic children aged 6–13.
3. Rappon et al. Control of myopia using diffusion optics spectacle lenses: 12-month results of a randomised controlled, efficacy and safety study (CYPRESS). Br J Ophthalmol. 2023 Nov;107(11):1709-1715.
4. Vision standards and requirements. Association of Optometrists. (n.d.). Date accessed 17th July 2025, from https://www.aop.org.uk/advice-and-support/clinical/vision-standards
5. Holden BA, Fricke TR, Wilson DA, et al. Global prevalence of myopia and high myopia and temporal trends from 2000 through 2050. Ophthalmology. 2016;123(5):1036-1042.
6. Xu L et al. High myopia and glaucoma susceptibility, the Beijing Eye Study. Ophthalmology. 2007;114(2):216-20.
7. Flitcroft DI. The complex interactions of retinal, optical and environmental factors in myopia aetiology. PRER 2013;31(6):622-60.
8. Younan C, et al. Myopia and incident cataract and cataract surgery: the blue mountains eye study. IVOS 2002;43(12):3625-3632.
9. Chen SJ, et al. Prevalence and associated risk factors of myopic maculopathy in elderly Chinese: the Shihpai eye study. IOVS 2012;53(8):4868-73.
10. Resolution: The standard of care for Myopia Management by Optometrists. World Council of Optometry. (2021, May 17). https://worldcouncilofoptometry.info/resolution-the-standard-of-care-for-myopia-management-by-optometrists Date accessed 17th July 2025. Reshaping care: New myopia management guidance released. AOA. (2021, January 12). https://www.aoa.org/news/clinical-eye-care/diseases-and-conditions/new-myopia-management-guidance-released American Academy of Ophthalmology Leads Global Initiative to address worldwide myopia epidemic. American Academy of Ophthalmology. (2020, November 11). https://www.aao.org/newsroom/news-releases/detail/academy-leads-initiative-to-address-myopia Date accessed: 7 August 2025.
11. Martínez-Pérez C, Villa-Collar C, Santodomingo-Rubido J, Wolffsohn JS. Strategies and attitudes on the management of myopia in clinical practice in Spain - 2022 update. J Optom. 2024 Jan-Mar;17(1):100496. doi: 10.1016/j.optom.2023.100496. Epub 2023 Oct 23.
12. SGV data on file 2022. Control of Myopia Using Peripheral Diffusion Lenses: Efficacy and Safety Study, 3-Year Report (n = 256, 14 North American sites).
13. Laughton D et al. Safety and efficacy of a novel spectacle lens for myopia control over three years. AAOptom conference 2022 presentation.
14. Wolffsohn JS, Hill JS, Laughton D, Hunt C, Young G. Visual Impact of Diffusion Optics Technology Lenses for Myopia Control. AAOptom conference poster presentation 2023.
15. Rappon J, Neitz J, Neitz M, et al. Two-year effectiveness of a novel myopia management spectacle lens with full-time wearers. Invest Ophthalmol Vis Sci 2022:e001790.
16. Laughton D., et al. Control of myopia using diffusion optics spectacle lenses in a Chinese population: 12-month results. 2025 ARVO poster presentation. 24-month, randomized and controlled clinical trial across 5 Chinese hospital sites, evaluating DOT spectacle lenses (n=128) versus control standard single vision spectacle lenses (n=58) in myopic children aged 6–13.
17. Hariton E, Locascio JJ. Randomised controlled trials - the gold standard for effectiveness research: Study design: randomised controlled trials. BJOG. 2018 Dec;125(13):1716.

